force and return promptly to its original condition
when the force is removed. These are the
properties of air that is used in pneumatic tires,
tennis balls and other deformable objects whose
shapes are maintained by compressed air.
KINETIC THEORY OF GASES
In an attempt to explain the compressibility
of gases, Bernoulli proposed the hypothesis that
is accepted as the kinetic theory of gases.
According to this theory, the pressure exerted by
a gas on the walls of a closed container is caused
by continual bombardment of the walls by
molecules of the gas.
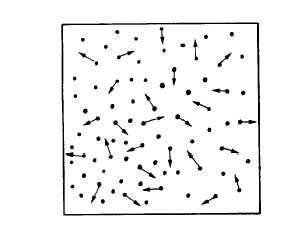
Consider the container shown in figure 11-2
as containing a gas. At any given time, some
molecules are moving in one direction, some are
traveling in other directions; some are traveling
fast, some slow, and some may even be in a state
of rest. The average effect of the molecules
bombarding each container wall corresponds to
the pressure of the gas.
As more gas is pumped into the container,
more molecules are available to bombard the
walls; thus the pressure in the container increases.
The gas pressure in a container can also be
increased by increasing the speed with which the
molecules hit the walls. If the temperature of the
gas is raised, the molecules move faster causing
an increase in pressure. This can be shown by
considering the automobile tire. When you take
a long drive on a hot day, the pressure in the tires
increases and a tire which appeared to be
somewhat “soft” in cool morning temperature
may appear normal at a higher midday tempera-
ture.
BOYLE’S LAW
When the automobile tire is initially inflated,
air which normally occupies a specific volume is
compressed into a smaller volume inside the tire.
This increases the pressure on the inside of the tire.
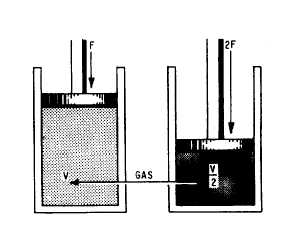
Charles Boyle, an English scientist, was among
the first to experiment with the pressure-volume
relationship of gas. During an experiment when
he compressed a volume of air he found that the
volume decreased as the pressure increased, and
by doubling the force exerted on the air he could
decrease the volume of the air by half. See figure
11-3. Recall from the example of the automobile
tire that changes in temperature of a gas also
change the pressure and volume. Therefore, the
experiment must be performed at a constant
temperature. The relationship between pressure
and volume is known as Boyle’s law. It states:
When the temperature of a gas is kept constant,
the volume of an enclosed gas varies inversely with
its pressure.
In equation form, this relationship may be
expressed as either
or
Equation 11-6
where V1 and P1 are the original volume and
pressure, and V2 and P2 are the final volume
and pressure (P1 and P2 are absolute pressures).
Figure 11-3.-Gas compressed to half its original volume by
Figure 11-2.—Molecular bombardment creating pressure.
11-4
a doubled force.